|
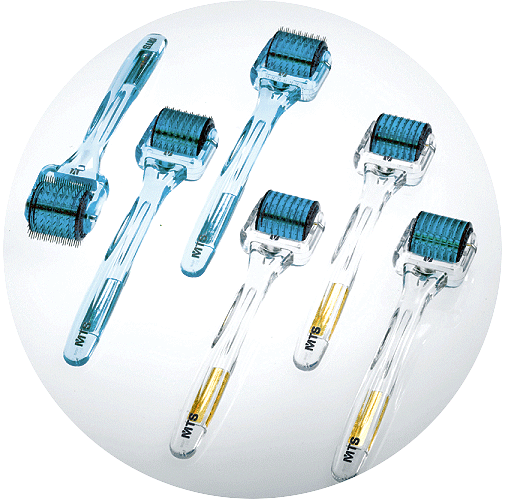
Microneedle Therapy System (MTS)-Roller Series
General Description
MTS Roller is a durable and high-quality precision tool. The barrel and handle are made from Lexan® polycarbonate resin. The axle is polished stainless steel axle. The 200 bristles (needles) – the highest count of any roller – are made from surgical stainless steel. A patented manufacturing process holds the needles permanently in place to within 0.02mm of the specified length from the roller base. All rollers have unique quality-control serial numbers.
| Type |
Model |
Length |
Methods
|
|

Personal Use
More Information
|
CR2
CR3
|
0.2mm
0.3mm |
Skin Restoration,
Improve Alopecia,
Homecare Therapy,
Active ingredient delivery |
|
|
CR5 |
0.5mm |
Skin Whitening,
Improve skin pigmentation,
Improve wrinkle (eye area),
Improve Alopecia,
Collagen Induction,
Drug delivery |
|
|
CR10
CR20
CR30 |
1.0mm
2.0mm
3.0mm |
Improve Wrinkle,
Collagen Induction,
Improve Skin Scar,
Cellulite treatment,
Skin Thickness,
Face Firming,
Face Body Lifting |
SPECIFICATION
- Roller Body: Diameter(20mm) X Width(21.5mm)
- Disc: 8 Microneedle discs
- Angle: 14.5°
- Needles Disc : 25ea (Total needles: 200 per each roller ubit)
- Material :
- High Quality Swedish Steel with Rustproof
- Special a method of construction
- 50% improvement of needle’s strength than a normal meterial
- Process:
Cutting, Grinder, Harden steel,
Extremely low Temper.
GUARANTEE: Gamma rays are emitted naturally by the sun, and are commonly known to cleanse and purify. Gamma irradiation is
a process commonly used by human-use device manufacturers to destroy bacteria and viruses. The flora and gamma notation used on the MTS Roller
labeling are symbolic of this, and our guarantee is that MTS rollers have been treated according to a validated gamma irradiation process,
by a certified supplier, to a standard, powerful level of 25.0kGY, for added assurance of initial cleanliness.
Storage and Use
Polycarbonate resin does not degrade from prolonged use or storage. Personal use rollers (CR2, CR3) should be replaced after about 6 months of regular use (2 to 3 times week). Clinical rollers (CR5, CR10, CR15, CR20) should be replaced after 10 applications. Rollers that have been dropped or bumped should be replaced immediately. Keep rollers in the protective case when not in use. For more information, consult CRL protocol guides.
Care of MTS Rollers™
Current MTS Roller models can withstand repeated immersion in “cold sterilization” agents such as germicides (phenol, ethyl alcohol) and Cidex™ (glutaraldehyde). Rollers should be completely dry before soaking to prevent dilution of the sterilization agent. Soak time depends upon the manufacturer's specifications. Rinse rollers thoroughly before using and return to its protective case after drying.
Next: Products | MTS Models | Personal Use- 
|